In times of medical ailment, individuals desperately seek medical attention — in particular, a cure or treatment to alleviate their illness. In these times of need, patients turn towards physicians for a diagnosis and effective treatment plan, relying on the latest technologies and therapeutic modalities to jumpstart a return to a normal lifestyle. However, what happens when there is no treatment, no cure and no therapy? When modern medicine no longer has anything to offer, patients may become afraid or desperate, and may become incredibly vulnerable.
There are many stories, and even more companies, which rely on the exploitability of patients in desperation. Companies may offer comprehensive “health care vacations” that allow patients to travel on an all-inclusive trip to a foreign country in order to receive medical treatments that are unapproved or even considered dangerous in their home country. This practice is known as medical tourism, and it has been exploited for centuries — in fact, the earliest incidence of medical tourism can be traced back thousands of years to Greek pilgrims who traveled across the Mediterranean to a place known as Epidauria in the Saronic Gulf seeking health from the healing god, Askiepios.
Since then, modern companies have offered medical therapies ranging from orthopedic surgery to stem cell transplantations to patients who either cannot afford the exorbitantly high health care costs in their own countries, or those desperate for a treatment. The practice of medical tourism may at times be warranted, such as in cases of financial limitations preventing access to proven treatments; however, in cases involving the acquisition of experimental or dangerous treatments, the exploitability of vulnerable patients for financial gain is lucidly unethical.
In 2013, an estimated 900,000 Americans went abroad for health care — with the number expected to rise in the coming years. This not only leads to decreased health care revenues in the United States, but also leads to potential dangers for the patients who might be seeking treatments that are considered too dangerous by the FDA or not approved in the United States for scientific reasons.
Ethically, the exploitation of patients by for-profit companies offering all-inclusive packages for medical therapies that are not approved in the United States has been widely debated. Some argue that it is the patients prerogative to seek the best health care available and that the government should not limit or restrict this right. Others argue that medical treatments which are offered in other countries and are deemed dangerous by the FDA means that companies can take advantage of vulnerable patients for financial gain.
In some cases, medical tourism can be financially justified — for example, “a standard knee replacement, which costs an average of $45,000 in the United States, can be performed for $6,500-$13,000 in India.” This provides individuals a financial incentive to receive their care abroad. However, what happens if follow-up care is needed, or the patient falls ill with an infection or complication from the surgery? The physician is many miles away and cannot care for their patient in this case, and the patient forfeits their right to sue for medical malpractice or negligence, thus increasing the likelihood of encountering massive amounts of medical bills without the possibility of receiving a settlement from the physician’s malpractice insurance company.
In other cases, individuals might seek experimental or controversial treatments, such as abortions, stem cell therapies or even sell/buy organs to the black market for financial gains. These are the ethically questionable practices of medical tourism, and many documented cases of damage incurred as a result of questionable medical treatments abroad have shed light on the dangers of experimental treatments abroad. These dangers include infection with various diseases due to contamination or inappropriate sanitation procedures, death from surgical procedures, increased risk of blood clots following the flight back, antibiotic resistance, and much more. In fact, the CDC’s website contains the following list of potential hazards patients may encounter when seeking health care abroad:
- Communication may be a problem. Receiving care at a facility where you do not speak the language fluently increases the chance that misunderstandings will arise about the care.
- Doctors may reuse needles between patients or have other unsafe injection practices, which can transmit diseases such as hepatitis and HIV.
- Medication may be counterfeit or of poor quality in some countries.
- Antibiotic resistance is a global problem, and resistant bacteria may be more common in other countries than in the United States.
- The blood supply in some countries comes primarily from paid donors and may not be screened, which puts patients at risk of HIV and other infections spread through blood.
- Flying after surgery increases the risk for blood clots.
These dangers do not even address the potential health hazards of receiving experimental treatments overseas. One of the most debated medical tourism topics currently, is the safety of stem cell therapies. According to the California Institute of Regenerative Medicine (CIRM), the “advancement of clinical therapies is best done in the setting of rigorous & formal clinical trials and in a structured regulatory framework.” This regulatory framework is sometimes circumnavigated or altogether avoided in many countries around the world. This “pseudoscientific” freedom presents the potential for physicians and scientists seeking financial, scientific, or clinical results to expose patients to therapies that have not been approved in the United States and may not be regulated to ensure patient safety. Although these treatments may be well intentioned, the lack of government oversight and rigorous testing significantly increases the chance of unforeseen long-term medical problems, such as pleiotropic teratomas from incompletely differentiated stem cell transplantations.
Although some governments may regulate the scientific endeavors of their researchers and physicians, the enforcement of the regulations may be lax — leading to substandard medical therapies and potentially dangerous outcomes for patients who are desperately seeking treatments for incurable illnesses, such as MS, pancreatic cancer or other autoimmune diseases. According to the FDA, unethical stem cell therapies can even be obtained in the United States — this can be gleaned from the thwarted attempts of three men who were arrested in the United States due to criminal activity “related to manufacturing, selling and using stem cells without FDA sanction or approval.” One of the individuals accused was a midwife who deceived mothers into banking their umbilical cord blood and then selling it to a lab that eventually purified the stem cells and used them for “unapproved stem cell procedures.”
The American government poorly regulates medical tourism perhaps out of respect for one of the guiding medical ethical principles, that of patient autonomy. However, this freedom may lead misguided or desperate patients into thinking they are receiving proven therapies by deceptive marketing strategies employed by medical tourism companies, when in fact, they are being exploited for financial gains by the corporations. One method of regulation could include ensuring that companies offering medical tourism opportunities to patients provide the patients with the scientific evidence supporting the therapies, which must be verified by the FDA in order for the company to be able to advertise to US patients. Any legislation that could help ensure the safety of patients who in times of illness, may be unable to logically reason the safety or danger of various treatments, is necessary since patients are currently being exploited by international companies for financial gains.
Interestingly, the United States at times benefits from medical tourism. Individuals from other countries with universal health coverage, sometimes travel to the United States in order to avoid extremely long wait times for certain medical procedures. Since these individuals cannot pay for the services in their home country, and must sometimes wait months for routine, non-life threatening medical tests. The wait time varies depending on the specialty, severity, and availability of health care providers — in Canada for example, the average wait time in 2013 for orthopedic surgery was nearly 40 weeks, and the average wait time to see a neurosurgeon was 17 weeks. These wait times can sometimes drive individuals to seek medical care in the United States, where individuals can sometimes pay cash for expedited appointments with specialists. The US medical system thus can sometimes benefit from medical tourism, however, in these cases, the patients are not being coerced by unfounded claims of cures for various incurable diseases.
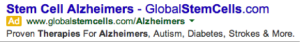
Physicians are bound by ethical principles that prioritize the safety and autonomy of their patient. As such, they are sometimes faced with the burden of presenting a patient with undesirable news. Physicians must provide patients with a realistic explanation of the disease, outcomes, therapies, and cannot overstate what medicine can provide. This could result in false hope by the patient and their family, which can place undue hardship that may exacerbate the negative associations of the disease process should those hopes not be met. This ethical balance of providing realistic hope is sabotaged by companies whom unwaveringly provide patients with hyped-up promises of cures and therapies for otherwise untreatable diseases. The following image was taken from a Google search for “stem cell therapy Alzheimer’s disease”:
This ad for a company known as “global stem cells” offers individuals with Alzheimer’s disease and their family members a false sense of hope that the disease can be treated with a “proven” stem cell therapy. Upon the realization that the companies unfounded promises cannot cure, but may even harm a patient with Alzheimer’s disease, both the patient and their family members may feel even more desperate. Unfounded promises made by companies may persuade patients to abandon their own physician, who may not be able to make as drastic of a promise of a cure as a for-profit company. This can result in an exacerbation of the underlying disease process, and may negatively impact the societal view of physicians and health care.
In summary, medical tourism is an emerging field that provides patients with an opportunity to receive health care abroad. Sometimes, going abroad to receive health care may be warranted, as in the case of an individual seeking a cheaper alternative to a proven and efficacious treatment. However, in certain cases, medical tourism isn’t warranted, and may at times take advantage of an individual with an untreatable disease by offering unfounded hopes of treatments or cures. In these cases, the ethical principles that normally guide the physician-patient relationship are not followed, and may result in undue harm towards the patient and his or her family. Legislation, which aims to regulate international medical tourism, may be justified in order to protect the safety of patients who may succumb to overhyped promises that are usually scientifically unfounded and potentially dangerous. Regulation would provide patients with a safer environment in which they can seek health care abroad. At times, patients may feel like they are undermining the authority or trust of their physician when they seek health care abroad. By regulating the process of medical tourism, it may allow patients to feel more comfortable speaking with their physicians about their international health care options, and may allow physicians the opportunity to warn patients of the dangers of unfounded stem cell therapies that claim to be curative. This would ideally result in a safer form of medical tourism, and may mitigate the ability for companies to make false promises and take advantage of a vulnerable patient population.